Educational Support for Mandatory Reporting of Serious Adverse Drug Reactions and Medical Device Incidents
Health Canada, ISMP Canada, Health Standards Organization (HSO) and the Canadian Patient Safety Institute (CPSI) have created 4 modules to explain and encourage reporting of serious Adverse Drug Reactions (ADR) and Medical Device Incidents (MDI) by hospitals, as mandated by the Protecting Canadians from Unsafe Drugs Act.
INTRODUCTION
The Protecting Canadians from Unsafe Drugs Act, also known as Vanessa’s Law, is intended to increase drug and medical device safety in Canada by strengthening Health Canada’s ability to collect information and to take quick and appropriate action when a serious health risk is identified.1 It will be mandatory for hospitals to report serious adverse drug reactions (serious ADRs) and medical device incidents (MDIs) to Health Canada, effective December 2019.2
A serious adverse drug reaction (serious ADR) is a noxious and unintended response to a drug that occurs at any dose and that
- requires in-patient hospitalization or prolongation of existing hospitalization,
- causes congenital malformation,
- results in persistent or significant disability or incapacity,
- is life-threatening, or
- results in death.3,4,5
A medical device incident (MDI) is an incident
- related to a failure of a medical device or a deterioration in its effectiveness, or any inadequacy in its labelling or in its directions for use, and
- has led to the death or a serious deterioration in the state of health of a patient, user, or other person, or could do so were it to recur.3,4,6
OVERVIEW OF REPORTING REQUIREMENTS
The mandatory reporting requirements for hospitals apply to therapeutic products, including pharmaceuticals (prescription and nonprescription drugs), biologic drugs, radiopharmaceutical drugs, disinfectants, and medical devices.3 Hospitals will be required to report a serious ADR or MDI to Health Canada within 30 calendar days of first documentation of the event within the hospital. Health Canada has specific definitions for these terms.
EDUCATIONAL MATERIALS
Health Canada, ISMP Canada, Health Standards Organization (HSO) and the Canadian Patient Safety Institute (CPSI) worked together to develop educational materials to support the implementation of mandatory reporting.
These materials provide information about the new regulatory requirements for mandatory reporting, describe reporting processes, offer strategies to promote and support reporting, and describe Health Canada’s review and communication of safety findings (Figure 1).
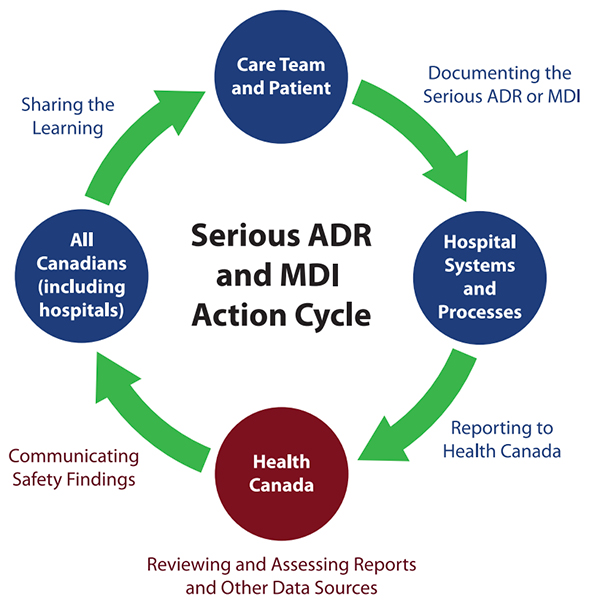
Figure 1. Conceptual model of serious ADR and MDI reporting by hospitals.7
There are 4 PowerPoint modules that contain core content intended for use by hospitals, health care professionals, educators, and patients and their families, to explain, describe, or promote the reporting of serious ADRs and MDIs. These materials (as entire modules or as individual slides or selected content) can be used for individual learning or incorporated into presentations, orientation, continuing education, and other information-sharing activities.
The modules were developed following a needs assessment and creation of an implementation plan. A pilot test was conducted over 3 weeks in March and April 2019 and garnered feedback from 255 of the pilot participants, who represented individual and organizational stakeholders from across Canada. The largest numbers of respondents were “pharmacists” and “hospitals”. Most respondents (83.1%) had reviewed all modules.
The following are key results from the pilot test:
- 89% of respondents reported that they had a good or strong understanding of the mandatory reporting requirements after reviewing the educational modules
- 94% indicated that using some or all of the slides would be a helpful approach to communicate information about Vanessa’s Law and reporting requirements
- 91% of respondents indicated that they planned to use the slides to communicate about the mandatory reporting requirements
- Many respondents provided helpful suggestions for additional information to be included in the final version of the PowerPoint modules.
- Respondents also provided suggestions for additional educational formats that could be considered for knowledge dissemination/translation in the future.
The educational modules were refined on the basis of the pilot feedback and Health Canada review. Updated modules are available from: https://www.patientsafetyinstitute.ca/en/tools Resources/Vanessas-Law/Pages/default.aspx
Module 1: Overview of Vanessa’s Law and Reporting Requirements
Module 2: Reporting Processes to Health Canada
Module 3: Strategies to Promote and Support Mandatory Reporting
Module 4: Health Canada’s Review and Communication of Safety Findings
The modules are intended to inform both health care providers and patients, to raise awareness of Vanessa’s Law, and to support reporting and learning. It is expected that individuals and organizations will be able to use these materials in various ways:
- Hospitals can include some or all of the content in information-sharing activities (e.g., “Lunch and Learn” sessions, presentations, orientation programs for staff).
- Educators in the health care sector can use the content in presentations or as part of a curriculum.
- Professional associations, societies, and regulatory colleges, as well as other training institutions for health care workers, may incorporate the content of the modules into accredited courses or continuing education certification programs.
- Patient and consumer organizations can help disseminate some or all of the information in the modules to increase awareness and knowledge among their members.
REPORTING AND LEARNING CONTRIBUTE TO THE SAFETY OF HEALTH PRODUCTS AND HEALTH CARE (Figure 2)
- Under Vanessa’s Law, the reporting of serious ADRs and MDIs to Health Canada is mandatory for hospitals.8
Note: Voluntary reporting of adverse reactions and medical device problems continues to be encouraged in all health care settings.
- Medication incident-related reporting and learning occurs through a separate and complementary program, the Canadian Medication Incident Reporting and Prevention System (CMIRPS).9,10
Analysis and shared learning from all of these reports contribute to continuous improvement in the safety of health products and health care.

Figure 2. Reporting and learning contribute to the safety of health products and health care.
![]()
The Canadian Medication Incident Reporting and Prevention System (CMIRPS) is a collaborative pan-Canadian program of Health Canada, the Canadian Institute for Health Information (CIHI), the Institute for Safe Medication Practices Canada (ISMP Canada) and Healthcare Excellence Canada (HEC). The goal of CMIRPS is to reduce and prevent harmful medication incidents in Canada.
Funding support provided by Health Canada. The views expressed herein do not necessarily represent the views of Health Canada.

The Healthcare Insurance Reciprocal of Canada (HIROC) provides support for the bulletin and is a member owned expert provider of professional and general liability coverage and risk management support.

The Institute for Safe Medication Practices Canada (ISMP Canada) is an independent national not-for-profit organization committed to the advancement of medication safety in all healthcare settings. ISMP Canada’s mandate includes analyzing medication incidents, making recommendations for the prevention of harmful medication incidents, and facilitating quality improvement initiatives.
Report Medication Incidents (Including near misses)
Online: ismpcanada.ca/report/
Phone: 1-866-544-7672
ISMP Canada strives to ensure confidentiality and security of information received, and respects the wishes of the reporter as to the level of detail to be included in publications.
Stay Informed
Subscribe to the ISMP Canada Safety Bulletins and Newsletters.
This bulletin shares information about safe medication practices, is noncommercial, and is therefore exempt from Canadian anti-spam legislation.
Contact Us
Email: cmirps@ismpcanada.ca
Phone: 1-866-544-7672
©2026 Institute for Safe Medication Practices Canada.
