Sharing Matters! Concerned Reporting Results in Improved Labelling to Reduce the Risk of Wrong-Route Errors
MedError.ca received a report from a consumer who almost ingested a boric acid vaginal suppository, mistaking it for an oral capsule. ISMP Canada shared this incident with Health Canada and the manufacturer, Szio+ Inc. Following discussions, the manufacturer has added clearer warnings to the outer box, bottle, and product insert.
INTRODUCTION
Boric acid vaginal products are available without a prescription to manage vaginal infections. MedError.ca (ISMP Canada’s consumer reporting program) received a report from a patient who almost ingested a boric acid vaginal suppository (Figure 1), mistaking it for an oral capsule. They shared their experience and provided suggestions to prevent a similar error. The following factors contributed to this near-miss event:
- use of a gelatin capsule that would typically be used for oral medications, vitamins, and supplements
- absence of a vaginal applicator (because an applicator can act as a cue to the route of administration)
- insufficient prominence of the route-of-administration labelling on the principal display panels

FIGURE 1. Original labelling and packaging for Utiva boric acid vaginal suppository product.
MANUFACTURER RESPONSE
ISMP Canada shared this incident with Health Canada and the manufacturer, Szio+ Inc. Following discussions, the manufacturer has added clearer warnings to the outer box, bottle, and product insert. These include a red bar on the outer box and insert stating “FOR VAGINAL USE ONLY. NOT FOR ORAL USE,” as well as a bottle cap safety seal that reads “DO NOT EAT” (Figure 2).
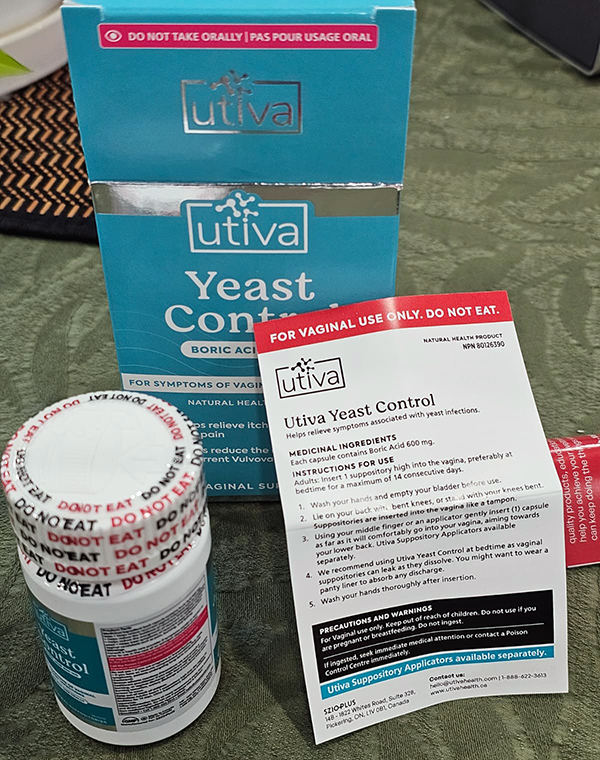
FIGURE 2. Improved labelling and packaging for Utiva boric acid vaginal suppository product.
RECOMMENDATIONS FOR MANUFACTURERS OF SUPPOSITORIES INTENDED FOR VAGINAL ADMINISTRATION
To reduce the risk of similar incidents, manufacturers are encouraged to do the following:
- Design vaginal suppository products in traditional suppository shapes (e.g., torpedo or cone-shaped) to distinguish them from oral medications.
- Display route-of-administration warnings prominently on the principal display panels of the outer box and the bottle (e.g., “For vaginal use only”).
- Include the formulation (e.g., vaginal suppository) directly under the ingredient name and strength on the principal display panels.
- Include a vaginal applicator with the product to support the correct route of administration.
- Add warnings on labelling and packaging with directions for the consumer if a suppository is mistakenly swallowed.
- Consult Health Canada’s Good Label and Package Practices Guide for Non-prescription Drugs and Natural Health Products when developing product labelling and packaging.
ISMP Canada is grateful to the consumer who submitted this report and greatly appreciates the commitment to patient safety demonstrated by Szio+ Inc.
![]()
The Canadian Medication Incident Reporting and Prevention System (CMIRPS) is a collaborative pan-Canadian program of Health Canada, the Canadian Institute for Health Information (CIHI), the Institute for Safe Medication Practices Canada (ISMP Canada) and Healthcare Excellence Canada (HEC). The goal of CMIRPS is to reduce and prevent harmful medication incidents in Canada.
Funding support provided by Health Canada. The views expressed herein do not necessarily represent the views of Health Canada.

The Healthcare Insurance Reciprocal of Canada (HIROC) provides support for the bulletin and is a member owned expert provider of professional and general liability coverage and risk management support.

The Institute for Safe Medication Practices Canada (ISMP Canada) is an independent national not-for-profit organization committed to the advancement of medication safety in all healthcare settings. ISMP Canada’s mandate includes analyzing medication incidents, making recommendations for the prevention of harmful medication incidents, and facilitating quality improvement initiatives.
Report Medication Incidents (Including near misses)
Online: ismpcanada.ca/report/
Phone: 1-866-544-7672
ISMP Canada strives to ensure confidentiality and security of information received, and respects the wishes of the reporter as to the level of detail to be included in publications.
Stay Informed
Subscribe to the ISMP Canada Safety Bulletins and Newsletters.
This bulletin shares information about safe medication practices, is noncommercial, and is therefore exempt from Canadian anti-spam legislation.
Contact Us
Email: cmirps@ismpcanada.ca
Phone: 1-866-544-7672
©2026 Institute for Safe Medication Practices Canada.
