Propofol 2% (20 mg/mL): Safety Considerations for Introducing a Novel Product into Hospitals
The increased worldwide demand for high-alert drugs used in the care of intubated and mechanically ventilated patients has heightened the risk of a shortage of these products. To address any shortage that does occur, an alternative product (typically the same drug in a different concentration, strength, packaging format, or dosage form) may be substituted for the medication that is unavailable.
Some Canadian hospitals will need to begin using an imported double-strength (2% or 20 mg/mL) propofol 100 mL product because there is a shortage of the 1% (10 mg/mL) propofol 50 mL and 100 mL products. To mitigate the risks associated with introducing this novel product into medication-use systems, the following preliminary considerations (from Table 1) are provided:
- Reserve the use of propofol 2% for maintaining sedation via continuous infusion in critical care areas (ideally only in the intensive care units) for adult patients who are mechanically ventilated.
- Consider if both the 1% (which continues to be available as a 20 mL vial) and 2% products are required in the same patient care area.
- Engage interdisciplinary staff in developing the implementation process, as a way to gain insight into critical aspects of providing safe care, including at transitions of patient care.
- Use medium- and higher-leverage strategies (e.g., alerts, forcing functions) in electronic systems (e.g., prescriber order entry, automated dispensing cabinets, infusion pump drug libraries).
- Create a go-live plan, including activation of communication and education plans and of updated drug libraries and alerts for infusion pumps.
- Supplement prescriber and staff education with information posters at the point of care in affected care areas.
- Only the propofol 1% product (20mL) should be drawn up into syringes; best practices for syringe labelling should be followed. It is envisioned that there will be no need to withdraw propofol 2% into a syringe.
- If the concentration of a propofol infusion needs to change (e.g., as a result of a patient transfer between care areas), prime new intravenous tubing with the new concentration.
INTRODUCTION
Drug shortages are one of the critical issues arising from the COVID-19 pandemic. The increased worldwide demand for high-alert drugs used in the care of intubated and mechanically ventilated patients has heightened the risk of a shortage of these products. To address any shortage that does occur, an alternative product (typically the same drug in a different concentration, strength, packaging format, or dosage form) may be substituted for the medication that is unavailable. The introduction of new products into the medication-use system must be carefully managed to mitigate risks, such as the potential for medication errors.1
REPORTED CONCERN
Due to an impending shortage of the propofol 1% (10 mg/mL) 50 mL and 100 mL products in Canada, some hospitals may need to use a double-strength (2% or 20 mg/mL) product that is now being imported from Europe (Figure 1). Propofol 2% is a product unfamiliar to Canadian practitioners and thus carries a risk of 2-fold overdose. A multipronged systems approach, including interprofessional communications and technical safeguards, is required to avoid errors with this product. Errors may also arise once the shortage is resolved and hospitals can again stock propofol 1% 50 mL and 100 mL products.

Figure 1. Propofol 2% 100 mL product imported from Europe under a Health Canada Interim Order (photograph courtesy of Fresenius Kabi Canada).
BACKGROUND
Health Canada has issued an Interim Order Respecting Drugs, Medical Devices and Foods for a Special Dietary Purpose in Relation to COVID-19 to expedite procurement and maintain an adequate supply of necessary medications and products during the pandemic. Patients with severe COVID-19 may require treatment, including intubation, in a critical care unit. Medications used to support patients in critical care include sedatives (e.g., propofol), analgesics (e.g., fentanyl), vasopressors (e.g., norepinephrine) and neuromuscular blocking agents (e.g., cisatracurium); these agents represent the majority of medications being imported under the interim order.2
The US Food and Drug Administration (FDA) is also authorizing importation of the European 2% product. The FDA has restricted propofol 2% for “maintaining sedation via continuous infusion in patients 16 years and older who require mechanical ventilation in an intensive care unit (ICU) during the COVID-19 pandemic.”3 While the product does not have the same restrictions in Canada, hospitals planning to use the propofol 2% product are considering the FDA guidance.
Propofol, a short-acting general anesthetic/sedative, is approved in Canada for sedation during intensive care and other procedures, as well as for induction and maintenance of general anesthesia.4 The demand for propofol is exceeding supply during the pandemic, and some hospitals have switched to alternative agents for specific indications. However, other hospitals that are treating a large number of intubated patients must have access to a continuous supply of propofol. The temporary importation of propofol 2% from Europe is intended to fill this need, but its introduction to the Canadian market carries certain risks. Not only is the 2% product double the strength of the 1% product familiar to Canadian practitioners, but it is also not currently supported by medication systems, including infusion pump drug libraries. A 2-fold overdose of propofol can result in hemodynamic instability, cardiovascular collapse, and death.
OPPORTUNITY FOR PROACTIVE SYSTEM RESPONSE
Several professional organizations, licensing bodies, group purchasing organizations, and governments have previously developed guidelines for managing drug shortages. Table 1 (PDF) consolidates concepts, human factors, and safety principles from these sources. The table highlights safety considerations for hospitals after a decision has been made to introduce the propofol 2% product into the medication-use system.
When implementing changes to support safe use of a new product, consider medium- and higher-leverage system-based strategies, such as alerts and forcing functions, to complement person-based interventions such as staff education and communications.5 As an example, Figure 2 depicts a concept clinical advisory alert that forces nurses to stop and acknowledge the product selected from the pump library. Fresenius Kabi Canada has developed a product alert poster. This poster can be placed in care areas to supplement existing education and other strategies.
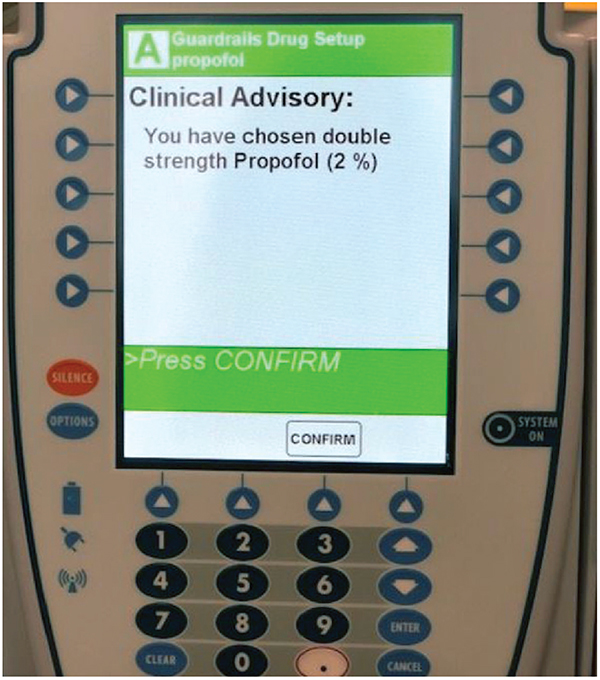
Figure 2. Example of a clinical advisory alert for infusion pump library (courtesy of Sinai Health).
If errors or near misses do occur, or if there are other concerns related to the adoption of the double-strength (2%) propofol product, they should be reported through the usual reporting mechanism, to Health Canada, and to the Canadian Medication Incident Reporting and Prevention System, to support further learning.
ISMP Canada invites all hospitals to share their implementation procedures and documents. These may be shared on the ISMP Canada website (with permission), to supplement the preliminary recommendations in this bulletin, for the benefit of other hospitals and practitioners. Please send documents and comments to info@ismpcanada.ca.
![]()
The Canadian Medication Incident Reporting and Prevention System (CMIRPS) is a collaborative pan-Canadian program of Health Canada, the Canadian Institute for Health Information (CIHI), the Institute for Safe Medication Practices Canada (ISMP Canada) and Healthcare Excellence Canada (HEC). The goal of CMIRPS is to reduce and prevent harmful medication incidents in Canada.
Funding support provided by Health Canada. The views expressed herein do not necessarily represent the views of Health Canada.

The Healthcare Insurance Reciprocal of Canada (HIROC) provides support for the bulletin and is a member owned expert provider of professional and general liability coverage and risk management support.

The Institute for Safe Medication Practices Canada (ISMP Canada) is an independent national not-for-profit organization committed to the advancement of medication safety in all healthcare settings. ISMP Canada’s mandate includes analyzing medication incidents, making recommendations for the prevention of harmful medication incidents, and facilitating quality improvement initiatives.
Report Medication Incidents (Including near misses)
Online: ismpcanada.ca/report/
Phone: 1-866-544-7672
ISMP Canada strives to ensure confidentiality and security of information received, and respects the wishes of the reporter as to the level of detail to be included in publications.
Stay Informed
Subscribe to the ISMP Canada Safety Bulletins and Newsletters.
This bulletin shares information about safe medication practices, is noncommercial, and is therefore exempt from Canadian anti-spam legislation.
Contact Us
Email: cmirps@ismpcanada.ca
Phone: 1-866-544-7672
©2026 Institute for Safe Medication Practices Canada.
