Standardize Units of Measure in the Expression of Levothyroxine Strength Across All Sources of Drug Information
ISMP Canada recently received a near-miss report involving a discrepancy between different units of measure for expressing the strength of levothyroxine. Opportunities exist for harmonization among sources of drug information, which would reduce the risk of dosing errors associated with miscalculation or conversion.
ISMP Canada recently received a near-miss report involving a discrepancy between different units of measure for expressing the strength of levothyroxine. A key contributing factor was the difference in how levothyroxine strength was presented in the local electronic health record (in micrograms) and in the patient profile within the provincial repository linked to the provincial formulary (in milligrams). This mismatch contributed to a conversion error and the subsequent preparation of a prescription containing tablets with 10 times greater than the intended strength.
Figure 1 illustrates the difference in how levothyroxine tablet strength is expressed in provincial/territorial drug formularies across Canada. Opportunities exist for harmonization among sources of drug information, which would reduce the risk of dosing errors associated with miscalculation or conversion.1,2
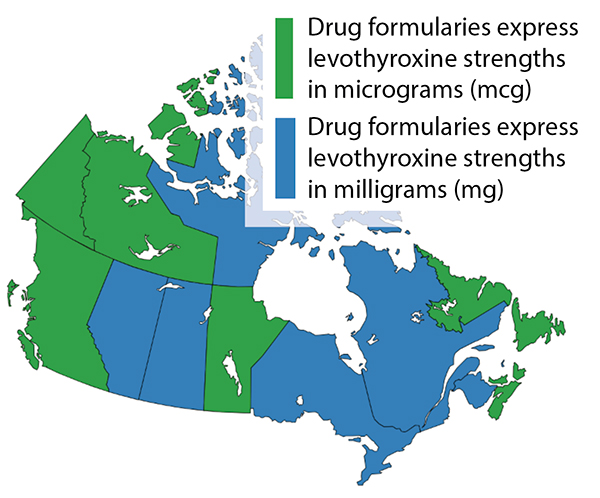
FIGURE 1. Distribution of provincial/territorial drug formularies that express levothyroxine strength in micrograms (mcg; green) and milligrams (mg; blue).
KEY RECOMMENDATION
Always display levothyroxine strengths and doses in micrograms (mcg), NOT in milligrams (mg).1
- This recommendation applies to all sources, including (but not limited to) provincial/territorial drug formularies, prescriptions, patient health records, pharmacy systems, group purchasing organizations’/wholesalers’ systems, medication administration records, and patient materials.1
- An important consideration and/or check is the display of medication information in the Canadian health product monograph. For example, the unit of measure is micrograms (mcg) in [human] levothyroxine product monographs from Health Canada’s Drug Product Database.3
![]()
The Canadian Medication Incident Reporting and Prevention System (CMIRPS) is a collaborative pan-Canadian program of Health Canada, the Canadian Institute for Health Information (CIHI), the Institute for Safe Medication Practices Canada (ISMP Canada) and Healthcare Excellence Canada (HEC). The goal of CMIRPS is to reduce and prevent harmful medication incidents in Canada.
Funding support provided by Health Canada. The views expressed herein do not necessarily represent the views of Health Canada.

The Healthcare Insurance Reciprocal of Canada (HIROC) provides support for the bulletin and is a member owned expert provider of professional and general liability coverage and risk management support.

The Institute for Safe Medication Practices Canada (ISMP Canada) is an independent national not-for-profit organization committed to the advancement of medication safety in all healthcare settings. ISMP Canada’s mandate includes analyzing medication incidents, making recommendations for the prevention of harmful medication incidents, and facilitating quality improvement initiatives.
Report Medication Incidents (Including near misses)
Online: ismpcanada.ca/report/
Phone: 1-866-544-7672
ISMP Canada strives to ensure confidentiality and security of information received, and respects the wishes of the reporter as to the level of detail to be included in publications.
Stay Informed
Subscribe to the ISMP Canada Safety Bulletins and Newsletters.
This bulletin shares information about safe medication practices, is noncommercial, and is therefore exempt from Canadian anti-spam legislation.
Contact Us
Email: cmirps@ismpcanada.ca
Phone: 1-866-544-7672
©2026 Institute for Safe Medication Practices Canada.
