A Multi-Incident Analysis of Reports Associated with Newer Insulins
Newer insulin products consist of biosimilars and faster-acting, longer-acting, or higher-concentration analogues. This safety bulletin describes a multi-incident analysis of reports involving newer insulin products and shares recommendations to enhance their safe use. Health care providers are encouraged to review their current processes and implement suggested improvements to support and promote the safe prescribing, transcribing, dispensing, and administration of insulin products.
INTRODUCTION
Over the past decade, several newer insulin products have been marketed in Canada. This safety bulletin describes a multi-incident analysis of reports involving newer insulin products and shares recommendations to enhance their safe use.
BACKGROUND
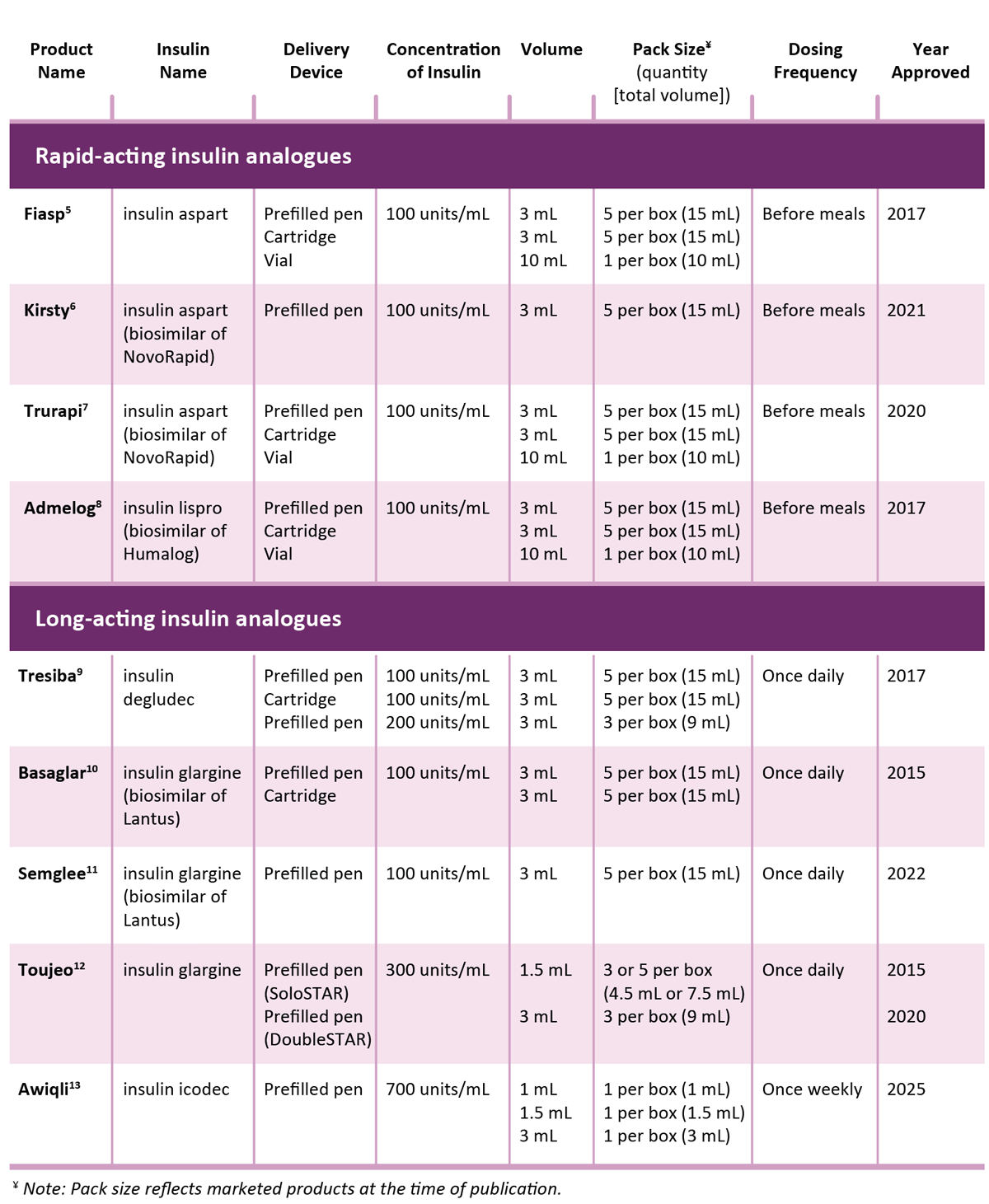
Newer insulin products (Table 1) consist of biosimilars* and faster-acting, longer-acting, or higher-concentration analogues.2 Insulin is a high-alert medication,3 so it is essential that health care providers stay informed to optimize patient safety in this evolving therapeutic landscape.
METHODOLOGY
Medication incidents associated with newer insulin products and submitted in the 5-year period between May 2020 and April 2025 were extracted from the Canadian Medication Incident Reporting and Prevention System (CMIRPS).† Key search terms included the product names of the newer insulin products (Table 1), as well as the names of the unique insulin analogues (such as icodec and degludec). The analysis was conducted according to the multi-incident analysis methodology outlined in the Canadian Incident Analysis Framework.4
TABLE 1. Characteristics of newer insulin products marketed in Canada and included in the analysis
QUANTITATIVE FINDINGS
A total of 1,136 incidents were extracted from the databases; 1,063 were included in the quantitative analysis‡ and 687 of those were included in the qualitative analysis. The remaining 376 incidents from the quantitative analysis were characterized as technical dispensing errors that did not elucidate safety concerns specific to newer insulin products; as such, they did not support further qualitative analysis.
The majority of incidents were classified by reporters as near misses (57%; n = 611) or incidents causing no harm (40%; n = 421), whereas 3% (n = 31) were reported to have caused harm.
Figure 1 shows the frequency of incidents reported for each of the top 5 medications, with insulin degludec (Tresiba) as the insulin product most frequently reported in incidents (n = 460). This finding may reflect its relatively longer time on the market and its designation as an open benefit across most provincial formularies.14 There were no reported incidents involving involving insulin icodec (Awiqli), the most recently approved long-acting insulin product; this absence of reports may reflect its relatively short time on the market.
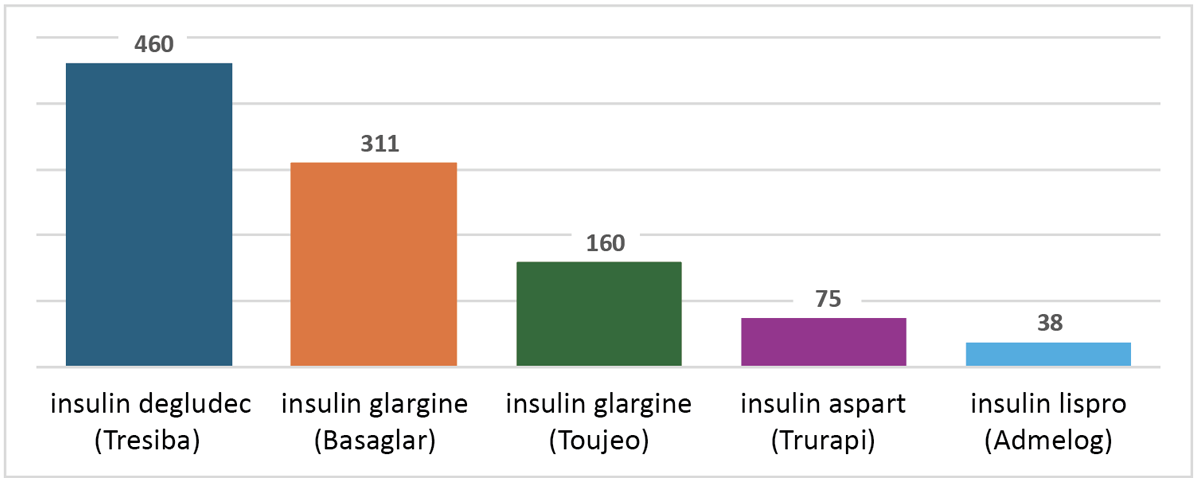
FIGURE 1. Number of incidents for each of the top 5 reported newer insulin products included in the quantitative analysis. Each incident may involve more than one insulin product. As described in the text, the frequency of reports for insulin degludec (Tresiba) may reflect the relatively longer time this drug has been in the market and its designation as an open benefit across most provincial formularies.14
QUALITATIVE FINDINGS
The qualitative analysis of incidents identified 3 themes and associated subthemes (Figure 2).

FIGURE 2. Themes and subthemes identified from analysis of reported incidents related to newer insulin products.
THEME: Vulnerabilities with Insulin Product Changes
Subtheme: Biosimilar clinical similarity versus interchangeability
Health care providers need to understand the difference between clinical similarity (a designation assigned by Health Canada) and interchangeability (a designation assigned by each provincial or territorial drug information database). Additionally, determining the reference biologic drug to which a biosimilar is clinically similar can be difficult, given that application of the same insulin name to multiple products does not indicate clinical similarity (e.g., insulin glargine in Basaglar is similar to insulin glargine in Lantus, but is not similar to insulin glargine in Toujeo) and given the existence of multiple biosimilars for a particular reference product (e.g., Trurapi and Kirsty are both biosimilars for NovoRapid [insulin aspart]).
Subtheme: Need for medication reconciliation
With the availability of newer insulin analogues and biosimilars, patients already receiving treatment for diabetes may bring prescriptions for new insulin products that are more convenient or affordable than their existing medications. However, if the patient’s medication profile is not updated regularly, and the insulin products are not reconciled after a switch to a biosimilar or another insulin product, there is an increased risk of errors.
Incident example: A patient’s previous prescription for Levemir (long-acting insulin) was inadvertently filled instead of the new prescription on file for Tresiba (long-acting insulin). The error was recognized when the patient questioned whether the product provided was the new insulin. Notably, the prescription for Tresiba documented it as a replacement for Levemir, but Levemir was not inactivated in the patient’s profile at the time the prescription was transcribed.
Recommendations for Prescribers
- Work with software providers to configure and regularly update prescribing software to designate applicable insulin products as “biosimilars” in a way that is visible during the prescribing process.
- Include both the brand/product and generic/insulin name on all insulin prescriptions.15,16
- Clearly specify the reference biologic for biosimilars (e.g., when there is a switch to another insulin) to help prevent confusion with other insulin products.
Recommendations for Pharmacy Teams
- Work with software providers to configure and regularly update pharmacy dispensing software to
- include newer insulin analogues and biosimilars
- designate applicable insulin products as “biosimilars” in a way that is visible during order entry and verification processes
- identify interchangeable insulin products or have an interface with a provincial drug information database that does so
- include information about duration of action in the product description
- alert users to potential duplication of the same type of insulin (i.e., duplicates of short-acting or long-acting insulins).
- Create a new entry for each new insulin prescription. Limit use of the copy function to prescriptions that are unchanged from the previous prescription in the patient’s profile.17
- Update the patient’s profile with each new prescription for insulin, and ensure that discontinued insulins, including previous dosing regimens, are inactivated.15
- Incorporate an independent double check into the verification process for insulin prescriptions, as they are high-alert medications.3,15
- Communicate any insulin prescription changes to the patient/caregiver both verbally and in writing (e.g., note attached to package for pick-up), so they know what to expect (e.g., different delivery device, concentration, or dose).18
THEME: Unfamiliar Insulin Product Characteristics
Many of the newer insulins have characteristics that differ from those of products already on the market, including multiple and/or higher concentrations, different delivery devices, and unfamiliar pack sizes. Although these variations offer more versatility in the management of diabetes, there is an increased risk of errors when health care providers are unaware of the differences.
Subtheme: Availability of multiple and high insulin concentrations
One of the most commonly reported types of errors was mix-ups between Tresiba U-100 (100 units/mL) and U-200 (200 units/mL) prefilled pens. Such mix-ups can have serious consequences due to the differences in concentrations and delivery devices. Tresiba U-100 delivers a maximum dose of 80 units in 1-unit increments, whereas Tresiba U-200 delivers a maximum dose of 160 units in 2-unit increments.11
Incident example: A patient had initially been using Tresiba 200 units/mL, but because of a drug shortage, they were switched to Tresiba 100 units/mL. The dose (in units) should have remained the same, but the patient was mistakenly told to double the dose. The following day, another pharmacy team member questioned the low inventory and determined that an unusually high number of boxes had been dispensed; this led to the error being recognized and remedied.
Subtheme: Variations in insulin pack sizes and delivery devices
Insulin products, including the newer insulin products, are available in various pack sizes (e.g., number of cartridges per box). Factors contributing to incidents with these products may include nonstandardized display of pack sizes in different pharmacy software systems (e.g., 5 prefilled pens containing 3 mL each may be displayed as “5” containers, “15” mL, or “1” box, and the particular display will affect quantity calculations). Insulin delivery devices also vary (e.g., prefilled pens, cartridges, and vials).
Incident example: A patient’s insulin order was incompletely transcribed on the medication administration record (MAR) as SoloSTAR 100 units/mL – 6 units subcutaneously with each meal. SoloSTAR is a type of insulin delivery device that can be used for multiple insulin products, but the specific insulin was not documented in the MAR. An incorrect insulin product was inadvertently selected and administered 9 times over 3 days before the incident was discovered.
Recommendations for Health Care Providers
- Make available educational materials (e.g., comparison chart) with key distinguishing characteristics for insulin products, including the brand/product and generic/insulin names, delivery devices, concentrations, volumes, and pack sizes.15
- Communicate insulin dosing in units (not mL) to reduce the risk of dosing errors and misinterpretation across products and concentrations.19
- Work with software providers to standardize display of the pack size of each insulin product in the quantity field.
- Incorporate an alert in the software to remind staff to confirm each patient’s insulin product and insulin delivery device, when applicable.
- “Show and tell” during patient counselling to demonstrate how to use the insulin product and delivery device to achieve the prescribed dose (in units), and use the teach-back method to ensure the patient’s/caregiver’s understanding.20,21 This process can allow for an additional check that the correct insulin product is being dispensed.
THEME: Refrigerator Storage Limitations
With an increasing number of insulin products, storage in the pharmacy’s refrigerator may become crowded, potentially impeding safe selection. Factors contributing to selection errors may include
- look-alike names and packaging for insulin products
- inadequate procedures for bundling and storing filled prescriptions of insulin products.
Incident example: A patient’s prescription for Tresiba was checked and placed in the fridge for pick-up, and another patient’s prescription for Humalog was checked and placed in the fridge for home delivery. The delivery driver was handed a number of packages, including the package containing Tresiba (instead of the one containing Humalog). The mix-up was discovered the next day when the first patient came to the pharmacy to pick up their Tresiba.
Recommendations for Pharmacy Teams
- Physically separate insulin product inventory in a way that prevents or lessens the chance of selection errors.
- Store short-, intermediate-, and long-acting formulations in individual bins or with shelf dividers.15,21
- Within each insulin grouping by duration of action, clearly separate products with concentrations greater than 100 units/mL, and add an auxiliary warning label as a reminder.
- Position insulin products in the refrigerator so that product labels (or patient-specific labels for filled prescriptions) are clearly visible.15
- Ensure that refrigerated storage space meets the pharmacy’s needs.
- Reassess and reconfigure refrigerator organization seasonally (e.g., after publicly-funded vaccine campaigns) to ensure adequate, clearly separated storage space for insulin and other cold-chain medications.
CONCLUSION
This analysis highlights vulnerabilities related to prescribing and dispensing insulin products, with a focus on the newer insulin analogues and biosimilars. Health care providers are encouraged to review their current processes and implement suggested improvements to support and promote the safe prescribing, transcribing, dispensing, and administration of insulin products.
![]()
The Canadian Medication Incident Reporting and Prevention System (CMIRPS) is a collaborative pan-Canadian program of Health Canada, the Canadian Institute for Health Information (CIHI), the Institute for Safe Medication Practices Canada (ISMP Canada) and Healthcare Excellence Canada (HEC). The goal of CMIRPS is to reduce and prevent harmful medication incidents in Canada.
Funding support provided by Health Canada. The views expressed herein do not necessarily represent the views of Health Canada.

The Healthcare Insurance Reciprocal of Canada (HIROC) provides support for the bulletin and is a member owned expert provider of professional and general liability coverage and risk management support.

The Institute for Safe Medication Practices Canada (ISMP Canada) is an independent national not-for-profit organization committed to the advancement of medication safety in all healthcare settings. ISMP Canada’s mandate includes analyzing medication incidents, making recommendations for the prevention of harmful medication incidents, and facilitating quality improvement initiatives.
Report Medication Incidents (Including near misses)
Online: ismpcanada.ca/report/
Phone: 1-866-544-7672
ISMP Canada strives to ensure confidentiality and security of information received, and respects the wishes of the reporter as to the level of detail to be included in publications.
Stay Informed
Subscribe to the ISMP Canada Safety Bulletins and Newsletters.
This bulletin shares information about safe medication practices, is noncommercial, and is therefore exempt from Canadian anti-spam legislation.
Contact Us
Email: cmirps@ismpcanada.ca
Phone: 1-866-544-7672
©2026 Institute for Safe Medication Practices Canada.
